Photodynamics of Bacteriorhodopsin
|
Bacteriorhodopsin (bR), a retinal protein in the superfamily of G-protein coupled receptors, is possibly the protein best studied by crystallography, which furnished not only high resolution structures of the protein at rest, but also detailed time-resolved images of various intermediates of its light-driven proton pump cycle. However, despite structural data resolved at the 1-2 Angstrom range we still do not know the basic mechanism of function of this protein. At the last conference on retinal proteins in Seattle in September 2002, after all the detailed time-resolved structures of bR were presented, the audience had to admit that we still do not know even such basic information as whether bR is a proton or a hydroxyl ion pump. Further progress seems to require knowing the dynamics of bR actually with a sub-Angstrom precision that is feasible only through quantum chemical and mechanical modeling, using the manifold observations as test of the theory. |
The first report of excited state dynamics of retinal
isomerization inside a protein
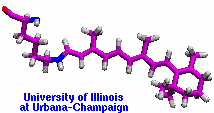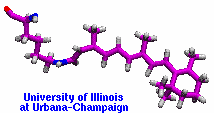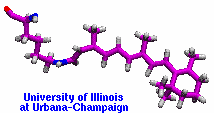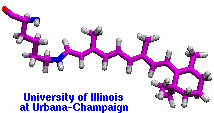 made with VMD |
Fortunately, the primary reactions in bR are induced by light and occur well within one picosecond, a short time that can be covered by the most advanced theoretical methods today, even if only barely so. Also fortunately, bR has been a focal point of attention of the best short time spectroscopy, having been in fact a driving force for femtosecond spectroscopy. No other protein has been probed by more advanced spectroscopic methods. As a result, theoretical descriptions are given ample opportunities for verification.
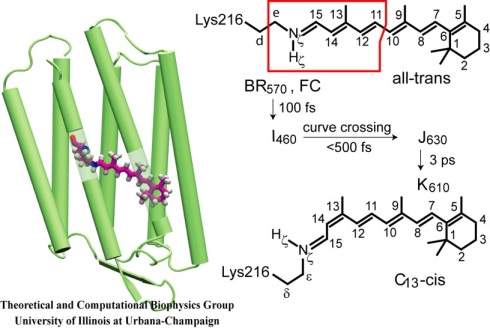
Very unfortunately, however, the dynamics of bR's primary reaction proceeds in an electronic excited state which poses an extreme challenge to quantum chemistry even in case of small molecules in vacuo. The latter have been studied for such type of reactions in a series of ground breaking papers that made scientific history. The time has come to apply the methodology to larger molecules and to reactions inside protein cavities with a coupling to actual protein motion. In a recent paper we have achieved such description for the first time.
In keeping with the great methodological advance that we achieved truly fundamental questions have been addressed in our study: Which degrees of freedom actually realize an isomerization (bond twist) event in a tight protein cavity? How directly or indirectly does the isomerization occur? How can the protein enhance the yield of the isomerization more than tenfold? How can the protein guide the reaction to a single product among many possible ones?
Lastly, our study is a splendid example of the convergence of the physical and biological sciences. The methods employed in the calculations and the experiments that supply confirmation belong to the most sophisticated kinds seen in the physical sciences today, yet they are applied to an exciting biological system that desperately requires the attention from the physical science.
Acknowledgement
This project is funded by grants MCB-9982629 and MCB- 02-34938 from the National Science Foundation.Related Projects:
Retinal Proteins team:



