Mechanosensitive channels
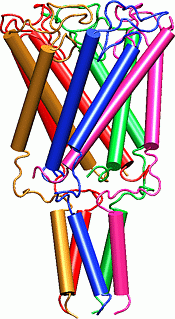
Mechanosensitive channels (MS channels) are proteins found in prokaryotic and eukaryotic cell membranes that open a conductance pore in response to mechanical stress. MS channels have been implicated in touch, hearing, cardiovascular regulation, sensing of gravity and osmotic stress. The bacterial large conductance MS channel (MscL) is thought to play an important role in rapidly regulating turgor pressure around the cell; studies have shown that when bacteria are challenged with a rapid osmotic downshock, many of the smaller cytoplasmic components are jettisoned into the medium, yet the bacteria remain viable. A permanent leak in the membrane, however, can eventually kill the bacterium. An understanding of the gating mechanism of bacterial MscL could thus lead to development of a new class of antimicrobial agents.

The MscL protein exhibits a high degree of primary sequence conservation within a group of bacteria which includes E. coli, on which most physiology experiments have been performed, as well as M. tuberculosis, from which the crystal structure was obtained. The determination of the crystal structure of MscL (Chang et al., 1998) revealed a protein with a homopentameric structure, approximately 50 Å wide in the plane of the membrane and 85 Å tall. Each 151-residue subunit consists of two transmembrane helices, labeled TM1 and TM2, and a cytoplasmic helix which extends some 35 Å below the membrane. The TMl helices are arranged so as to block diffusion through the channel at their N-terminal ends; this region of the protein also exhibits very high sequence conservation. A loop region between TM1 and TM2 extends into the pore, which may also contribute to the conductance of the channel. Excision of the cytoplasmic domains has been found to have little effect on the gating properties of the channel (Ajouz et al., 2000).
Equilibrium Simulation
Results of the simulations of the protein in the full membrane-water system reflect a protein that is quite stable in the closed state. This is to be expected from patch-clamp data (Sukharev et al, 1999), which reveal a channel with zero conductance until significant tension is applied. Large-scale changes in the shape of the protein could not be expected during the progress of a 3~ns simulation; it is, therefore, possible that a much longer simulation could reveal a somewhat different closed state. We believe that we have described the essential features of this protein on the time scale of several nanoseconds, and find encouraging correspondence with experiments.
Fluctuations on the scale of individual residues were found to be in good agreement with corresponding measurements from ESR experiments, confirming the validity of our protein model. Water penetration in the pore was found to extend only to hydrophilic residues, i.e., only as far as Thr25. This result lends support to proposed mechanisms of MscL gating that postulate a change in the solvent environment of hydrophobic residues in the pinched region of the protein during gating.
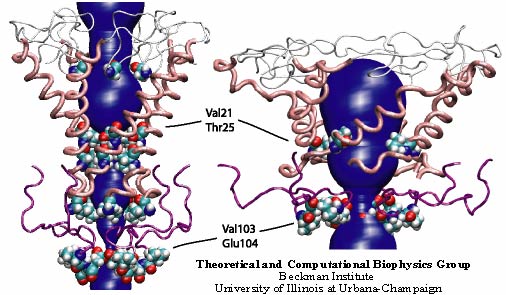
Applied Surface Tension Simulations
Though a realistic simulation of MscL must include the membrane and surrounding water, we can investigate the mechanics of the protein itself without these external media. To this end we conducted a series of simulations of the same protein structure as in the membrane simulations, but with no membrane or water present. The simulations were conducted at constant surface tension and zero normal pressure. To our knowledge, this is the first time surface tension has been used in molecular dynamics simulations to elicit a conformational change in a protein.
Our simulations of the bare protein using an applied surface tension to induce conformational change provided remarkably consistent results: the protein retained its secondary structure while radically reforming its tertiary structure to form a large pore. Retention of secondary structure was an important validity check since the native lipid environment would not have allowed alternative hydrogen bonds to form. The observation that the transmembrane helices flattened out corresponds well with recent measurements made of the effect of membrane thickness on MscL gating (Kloda and Martinac, 2001). In these measurements, it was found that when MscL was placed in a thinner membrane, it remained for a longer period of time in its open state. This would seem to suggest that the open conformation of MscL is flatter than the closed structure.
Application of Pressure Profiles
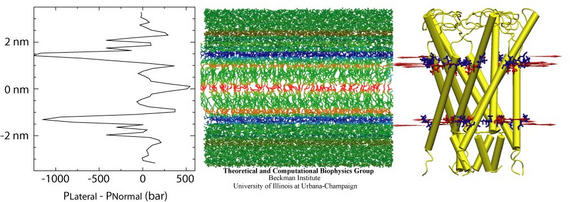
In order to study the protein-lipid interactions leading to gating that were neglected in our earlier work, we initially placed Eco-MscL in a fully hydrated lipid bilayer, with sufficient lipid to form a bulk phase far from the protein. We then attempted to gate the channel by applying moderate (50 dyn/cm) amounts of surface tension to the entire system, as was done in our earlier applied surface tension simulations. These attempts failed because the membrane stretched and compressed, while maintaining nearly constant volume, well before MscL's conformation could be affected. This result was, in retrospect, not surprising, given that the area compressibility of lipid bilayers is much smaller than the volume compressibility.
An alternative approach is to instead model the pressures that would be present in a stretched bilayer, rather than simulate the stretching of the bilayer itself. In order to gauge the extent to which the applied forces used in our simulations mimics the effect of an actual lipid bilayer, we calculated the lateral pressure in a DLPE bilayer as a function of depth in the membrane. The pressure profile is very similar to that obtained by Lindahl and Edholm in their 2000 paper. By far the most prominent feature of the pressure profile is the negative lateral pressure peak just below the lipid headgroups. This peak corresponds to high surface tension: the membrane would like to reduce its surface area at this depth. In the center of the membrane are three smaller, positive peaks corresponding to forces that tend to increase the membrane area at this depth. We thus conclude that the membrane may be mimicked by radial forces near the lipid-water interface, with compressive forces in the transmembrane part and just outside the lipid bilayer.
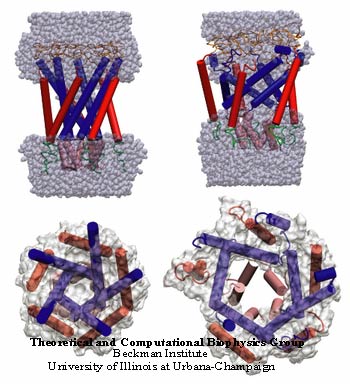
Steered Molecular Dynamics Studies
Results of our SMD simulations are described here. Using this force protocol derived from pressure profile simulations, we obtained an expanded state of the MscL channel from a steered molecular dynamics trajectory. In this series of simulations, water was present but no membrane. Starting structures are available here, in the form of a psf and pdb file. After 10 ns the pore radius was 8.2 Angstroms. Expansion of the transmembrane region took place in conjunction with a tilting of the inner helices toward the membrane plane. Outer helices tilted to a much smaller extent, but did expand outward as the pore opened. At the same time, neither the periplasmic loops nor the N-terminal helix bundle opened appreciably. The secondary structure of the transmembrane helices was somewhat degraded on their periplasmic ends, but the secondary structure near the tightest constriction of the pore, where the M1 helices meet, was quite sound for the first 10 ns of simulation O4 before the applied forces disrupted the structure. It is also clear from the figure that water completely wetted the pore in the expanded state of the channel, but did not enter the region where a lipid bilayer would normally be found. This can be attributed to the affinity of the water for the hydrophilic parts of the structure, as well as to the periodicity of the simulation along the channel axis, which permitted the water to form a continuous bulk region in the cytoplasmic and periplasmic regions of the channel.
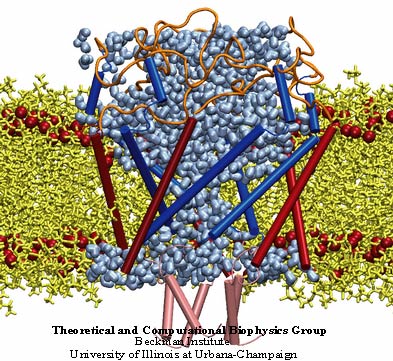
An SMD simulation of MscL in an explicit lipid environment was also performed to check that the simulations described above were not unduly affected by the absence of a bilayer. The initial force applied to the channel residues was 70 pN, just as in the simulation described above. In the simulation without membrane, there was significant expansion of the transmembrane pore constriction within 5 ns; in contrast, in the full membrane simulation the pore did not open at all within the first 6.6 ns, likely due to the bilayer pressing against the outside of the channel. The applied force on the cytoplasmic residues was therefore increased in steps to accelerate the gating process; the surface tension was also increased to decrease the extent to which membrane pressure held the protein shut. Within 6 more nanoseconds the first stages of gating were observed. Comparison of the two simulations shows that the water distributions around the cytoplasmic and periplasmic sides of the channel in the two simulations are virtually identical, suggesting that there is no unrealistic hydration of the hydrophobic exterior of MscL present when no membrane was included.
MscL Gating Model
The mechanism of MscL gating from the closed state to the expanded state may be summarized as follows. Initial expansion of the channels begins in the periplasmic ends of the transmembrane helices (residues 30-45 and 75-85). No serious energy barriers are encountered in this phase of the expansion, as evidenced by the fact that even SMD simulations with small forces managed to induce this level of expansion. The outer transmembrane helices may expand as much as 6 Angstroms from the closed state without necessarily opening the hydrophobic constriction at Leu19 and Val23. No further expansion appears to be possible without opening of this first gate. The mechanical tension necessary to open this gate should come from the cytoplasmic side of the bilayer.
Progress towards expansion of the hydrophobic constriction appears to require Ala20 slipping past a pocket formed by Ile25 and Phe29 in a neighboring subunit; all three of these residues are very well conserved among MscL homologs. Once this slippage has occurred, expansion of both inner and outer transmembrane helices can continue; transmembrane helices from neighboring subunits expand together due to both close packing and a stable salt bridge between Lys31 and Asp84.
The opening of the first gate is quite decoupled from the expansion of the N-terminal helix bundle: the average radius of residues 16-28 increased by over 10 Angstroms, while residues 1-13 did not expand at all. The flexibility in the linker region of MscL comes entirely from Gly14; Arg13 and Asn15 remained in the allowed portion of Ramachandran space and did not become excessively distorted to allow the expansion of the inner transmembrane helix.
Click here for a 2.8M movie of the channel opening.
Publications
Investigators
Related TCB Group Projects
Page created by Justin Gullingsrud and maintained by Marcos Sotomayor.



