ATP Synthase
|
The membrane unit Fo converts the transmembrane electrochemical potential into mechanical energy that drives the central stalk rotation inside the solvent-exposed F1 unit. The rotation causes cyclic conformational changes in the F1 unit catalytic sites, which drive ATP synthesis (the "binding change" mechanism). ATP synthase can also operate in the reverse direction, utilizing the energy released in the process of ATP hydrolysis to pump protons across the membrane. Several studies of this system are underway:
|

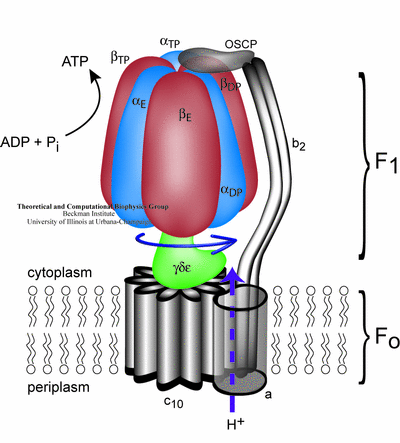 Living cells depend on an efficient transformation of the energy
derived from light and foodstuff into the chemical energy of adenosine
triphosphate (ATP), the universal energy carrier.
The enzyme that
facilitates this type of energy conversion in bacteria, chloroplast
and mitochondria is ATP synthase, a complex of two molecular motors
mechanically coupled by a common central stalk.
Living cells depend on an efficient transformation of the energy
derived from light and foodstuff into the chemical energy of adenosine
triphosphate (ATP), the universal energy carrier.
The enzyme that
facilitates this type of energy conversion in bacteria, chloroplast
and mitochondria is ATP synthase, a complex of two molecular motors
mechanically coupled by a common central stalk.


