Vibrationally assisted olfaction
The sense of smell
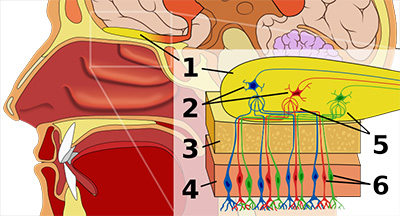
Figure 1. Human olfactory system. 1: Olfactory bulb 2: Mitral cells 3: Bone 4: Nasal epithelium 5: Glomerulus (olfaction) 6: Olfactory receptor cells. The image adapted from here.
Olfaction is the sense of smell. This sense is mediated by specialized sensory cells of the nasal cavity of vertebrates. Many vertebrates, including most mammals and reptiles, have two distinct olfactory systemsthe main olfactory system, and the accessory olfactory system (used mainly to detect pheromones). For air-breathing animals, the main olfactory system detects volatile chemicals, and the accessory olfactory system detects fluid-phase chemicals [Hussain, Saraiva, Korsching, PNAS 106: 4313 (2009)]. Olfaction, along with taste, is a form of chemoreception. The chemicals themselves that activate the olfactory system, in general at very low concentrations, are called odorants. Volatile small molecule odorants, non-volatile proteins, and non-volatile hydrocarbons may all produce olfactory sensations. Some animal species are able to smell carbon dioxide in minute concentrations.
The lock-and-key theory of olfaction
The mainstream theory of olfaction is based on the so-called lock-and-key model [Silverman, The Organic Chemistry of Enzyme-Catalyzed Reactions, Academic Press, 2002]. According to this theory, size, shape and functional groups of odorant compounds determine activation of olfactory receptors. Once an associated odorant molecule binds to an olfactory receptor, the receptor is activated and triggers a neural signal. Olfactory receptors have been identified as G-protein coupled receptors (GPCRs) [Buck and Axel, Cell (Cambridge, Mass.) 65, 175 (1991)]. Since most of the GPCRs, e.g., β-adrenergic agonists, are activated through binding of ligands and are highly sensitive to ligands' conformation and surface properties, the lock-and-key mechanism seems to be a natural explanation of olfaction.
However, the lock-and-key mechanism as the sole discriminant was questioned after a sensory analysis of isotope effects of benzaldehyde and its derivatives (C6(ring)-benzaldehyde, CHO-benzaldehyde and benzaldehyde-d6) acting as odorants [Haffecden, Yaylayan, Fortin, Food Chem. 73, 67 (2001)]. Based on the duotrio test by 30 trained panelists, the authors reported a significant statistical odor difference of benzaldehyde-d6 relative to non-deuterated benzaldehyde, evoking a shift of vibrational frequencies in the benzaldehyde samples. Moreover, it was demonstrated recently that Drosophila melanogaster can distinguish between deuterated and regular odorant compounds, despite the fact that the shapes of such compounds are identical, while the only difference arises in the vibrational spectrum [Franco, Turin, Mershin, Skoulakis, PNAS 108, 3797 (2011)].
Vibrationally assisted mechanism of olfaction
An alternative mechanism of olfaction had been suggested [Dyson, Chem. Ind. 57 647 (1938); Wright, J. Theor. Biol. 64, 473 (1977)]. According to this mechanism the differences in key vibrational frequencies of odorant compounds contribute to odor perception [Turin, Chem. Senses 21, 773 (1996)]. A plausible way of sensing the vibrational spectrum of an odorant is furnished through electron transfer in the olfactory receptor [Solov'yov, Chang, Schulten, 2012]. The mechanism is schematically illustrated in Fig. 2.
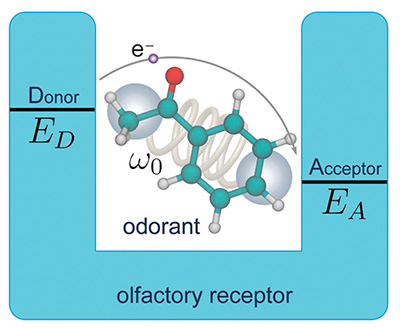
Figure 2. Principle of vibrationally assisted olfaction. The odorant molecule with characteristic frequency ω0 binds to the olfactory receptor binding pocket forming an electrondonoracceptor complex with donor energy ED and acceptor energy EA. Electron tunneling from the donor site to the acceptor site of the olfactory receptor is enhanced if the vibrational frequency of the odorant molecule matches the energy difference Δε=ED-EA.
see also movie, 2.4MB
Initially, an electron resides on the electron donor (D) site of the olfactory receptor. The receptor carries also an electron acceptor (A) site, but the D and A site redox energies differ by an amount Δε, too large to facilitate thermally assisted electron transfer on a relevant time scale, i.e., hundreds of nanoseconds or shorter, without the presence of an odorant. However, once a suitable odorant binds to the receptor, the vibrational frequency of the odorant that matches Δε makes electron transfer possible, as the energy Δε can be released to the vibrational mode of the odorant. Odorant vibrations employed that way have particularly high frequencies. Thus, the key idea of the vibrationally assisted electron tunneling mechanism is to increase the tunneling rate in the olfactory receptor through vibrational excitation of the odorant, to make it larger than the elastic tunneling rate in the olfactory receptor, a rate that by itself is too small for olfactory reception. We like to emphasize that the stated mechanism complements, but does not replace, a lock-and-key mechanism of olfaction, i.e., both shape and vibrations should be significant for odor perception.
Theory of vibrationally assisted electron transfer
Electron transfer between donor and acceptor sites of an olfactory receptor (see Fig. 2) can be described through a model Hamiltonian H
which accounts for the essential physical interactions in the system. Here HR denotes the olfactory receptor Hamiltonian, HRO describes the coupling interaction between the olfactory receptor and an odorant molecule, HRenv describes the coupling of the olfactory receptor to the thermal environment, and HT accounts for electron tunneling from donor to acceptor sites of the olfactory receptor. Thus, the Hamiltonian H describes the entire olfactory system which consists of the receptor, odorant, and molecular environment.
In the vibrationally assisted mechanism of olfaction, the olfactory receptor is a quantum mechanical system with two energy levels, which correspond to the electron donor and acceptor states. In [Solov'yov, Chang, Schulten, 2012] we have elaborated that Eq. (1) allows to determine the vibrationally assisted electron tunneling rate from the donor to acceptor site of the olfactory receptor
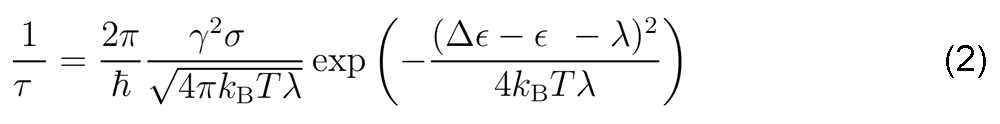
Here γ is the hopping integral between the donor state and the acceptor state of the olfactory receptor; σ is the so-called HuangRhys factor which describes the inelastic nature of the electron tunneling process due to the coupling between the olfactory receptor and odorant molecule; The reorganization energy λ describes the coupling of the olfactory receptor donor and acceptor states to the vibrations of the molecular environment; Δε represents the energy difference between the olfactory receptor donor and acceptor states (see Fig. 2); Finally, ε is the energy of the odorant excited state gained through electron transfer accounting also for coupling to the odorant vibration.
Tunneling rate in the olfactory receptor
To characterize vibrationally assisted tunneling, the tunneling rate was calculated as a sum over individual rates for all vibrational modes of the odorants. Thus, the contour plots in Fig. 3 show the total electron tunneling rate 1/τ calculated as a function of the energy difference Δε and the reorganization energy λ for acetophenone, citronellyl nitrile and octanol. The typical times for intrinsic electron tunneling in biological systems occurring on a length scale of 515 Angstroems are 10100 ns. Accordingly, Fig. 3 shows only rates for which tunneling occurs faster than 100 ns.
| Figure 3. Electron tunneling rates in an olfactory receptor. Contour plots showing the calculated electron tunneling rates as a function of energy difference Δε and the reorganization energy λ for (a) acetophenone, (b) citronellyl nitrile and (c) octanol. For convenience the color-code indicates the characteristic tunneling times of the electron in the receptor, i.e., the inverse of the tunneling rate. The rates of the regions enclosed by the dashed rectangles are shown in Fig. 4 in greater detail. The faded colors indicate regions where the inelastic tunneling rate 1/τ falls below the elastic tunneling rate, 1/τ0. | 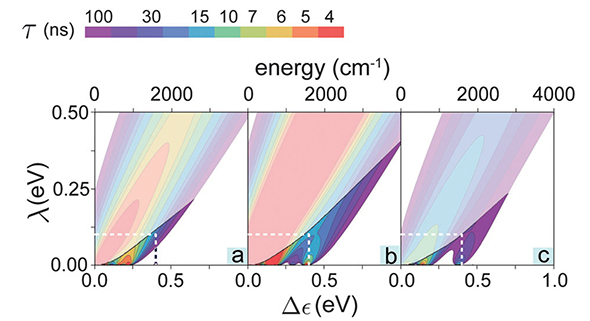 |
The rates 1/τ for the three odorants studied, shown in Fig. 3ac, behave similarly, although the maximal tunneling rates are slightly different. However for reorganization energies λ≥0.5 eV, the tunneling rate shows only a weak dependence on the energy difference Δε, with a single peak at Δε≈0.5 eV for all studied odorants. This λ-range renders then the vibrationally assisted mechanism impractical for resolving different odorant compounds, as all of them will act similarly on the receptor.
Figure 4 illustrates the differences in 1/τ calculated for acetophenone, citronellyl nitrile and octanol presenting 1/τ evaluated in the (λ, Δε)-range [0 ≤ λ ≤ 0.1 eV, 0 ≤ Δε ≤ 0.4 eV]. The 1/τ maxima at certain Δε values are related to the characteristic vibrational frequencies of the odorants. Thus, for example, citronellyl nitrile vibrations labeled 2 and 3 in Fig. 4b correspond to the maxima of the electron tunneling rate at Δε2=0.29 eV and Δε3=0.38 eV, respectively. Further inspection of Fig. 4 reveals that the energy dependence of the tunneling rate reflects the key IR active vibrations for the three studied odorants and, obviously, recognition of the odorants through the vibrationally assisted electron tunneling mechanism is feasible.
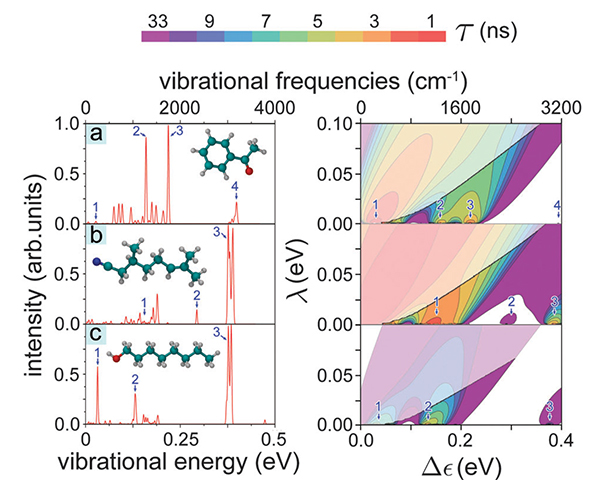 |
Figure 4. IR spectra and electron tunneling rates. (right) Contour plots of the calculated electron tunneling rates shown as a function of the energy difference Δε ≤ 0.4 eV and reorganization energy 0 ≤ λ ≤ 0.1 eV for (a) acetophenone, (b) citronellyl nitrile and (c) octanol. The rates shown are taken from Fig. 3. For convenience the color-code indicates the characteristic tunneling times of the electron in the receptor, i.e., the inverse of the tunneling rate. The faded colors indicate regions where the inelastic tunneling rate 1/τ falls below the elastic tunneling rate 1/τ0. (left) Calculated normalized IR spectra for the three odorants with structures shown in the insets. Numbers 1, 2, 3, 4 label in the left and right panels corresponding IR vibrations. |
Isotope effect
Deuteration affects the vibrational frequencies of odorants due to substitution of hydrogen by the heavier deuterium. Deuteration offers an important test for the vibrationally assisted mechanism of olfaction as deuteration does not affect the surface properties of odorants. The electron tunneling rates for three deuterated odorants (acetophenone, citronellyl nitrile and octanol) were calculated. The dependencies of the electron tunneling rate on Δε and λ are shown in Fig. 5.
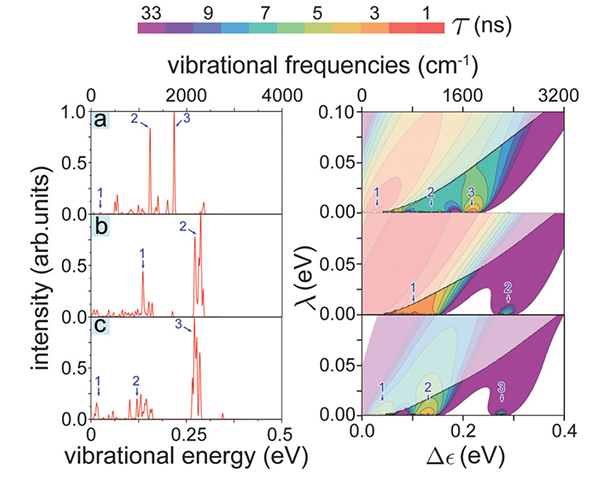 |
Figure 5. IR spectra and electron tunneling rates for deuterated odorants. IR spectra and electron tunneling rates calculated for deuterated (a) acetophenone, (b) citronellyl nitrile and (c) octanol. The presentation is analogous to that in Fig. 4. |
An important result is observed for one particular vibrational mode in deuterated octanol (see Fig. 5c) and non-deuterated citronellyl nitrile (see Fig. 4b), where the increase in the electron tunneling rate occurs in either case at around Δε ∼ 0.29 eV (see peak 2 in Fig. 4b and peak 3 in Fig. 5c). The match of the electron tunneling rate at a certain vibrational frequency for two different odorants suggests that both odorants could activate the same olfactory receptor, namely the one tuned to detect this specific vibration, as long as both odorants have the proper shape. The enhancement of the tunneling rate at 0.29 eV (vibrational frequency of 2257 cm-1) for deuterated octanol and non-deuterated citronellyl nitrile is in agreement with a recent experimental study performed on Drosophila melanogaster [Franco, Turin, Mershin, Skoulakis, PNAS 108, 3797 (2011)], where the authors reported that fruit flies react differently to natural octanol and citronellyl nitrile, while they show similar behavior if exposed to deuterated octanol and non-deuterated citronellyl nitrile.
Odorant discrimination through shape and vibrational frequencies
Figure 6 demonstrates the principle of odorant discrimination through shape and vibrational frequencies and, in particular, illustrates the similarity between deuterated octanol and non-deuterated citronellyl nitrile. Shape is a decisive factor to link odorants to a particular receptor. Figure 6 illustrates two types of odorant receptors, chosen to detect either octanol (light blue cones) and citronellyl nitrile (pink cones). Since the size of octanol and citronellyl nitrile is similar (the molecules have a length of about 11 Angstroems, and are about 34 Angstroems in diameter), both receptors could likely accommodate either molecule. Thus, the octanol and citronellyl nitrile can be recognized only through the vibrationally assisted electron transfer mechanism if the octanol (citronellal nitrile) receptor exhibits a Δε value characteristic of an octanol (citronellal nitrile) vibrational energy quantum, thereby enhancing the electron tunneling rate in the octanol receptor through the presence of octanol, and similarly in the citronellyl nitrile receptor through the presence of citronellyl nitrile. Figure 6 illustrates also how electron transfer through the receptor is affected by deuteration. Deuteration of octanol decreases the vibrational frequency of some vibrational modes, and certain modes become energetically close to a vibrational mode in citronellyl nitrile. This results in the mismatch of the vibrational energy εoctdeu with Δε of the octanol receptor, but in a match of this energy to the Δε value of the citronellyl nitrile receptor such that deuterated octanol activates the citronellyl nitrile receptor.
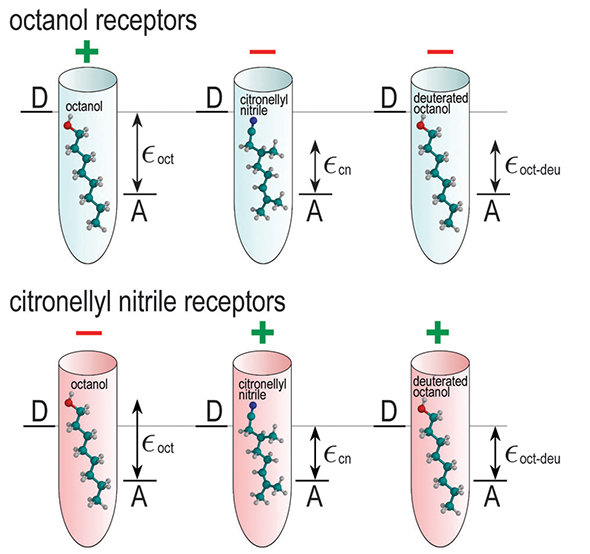 |
Figure 6. Discrimination of deuterated odorant. Illustrated is here the principle of odorant discrimination through shape and vibrational frequency. Octanol receptors (light blue) and citronellyl nitrile receptors (pink) accommodate octanol, citronellyl nitrile, and deuterated octanol. Since the shape of octanol and citronellyl nitrile is similar, both odorants fit into either receptor. The receptors possess different Δε, i.e., the energy between the donor state D and acceptor state A, tuned to a certain vibration of the odorant. The ability of a receptor to respond to odorant binding is denoted with a green "+" sign, while the red "-" sign shows that the receptor is passive to the corresponding odorant binding. The characteristic vibrational energies of octanol, citronellyl nitrile, and deuterated octanol are denoted εoct, εcn, and εoctdeu, respectively. |
More generally, Fig. 7 illustrates how deuteration affects the olfactory discrimination landscape by shifting the odorant-specific activation domains along the odorant vibrational excitation coordinate (blue domain). The figure illustrates that in some cases deuteration leads to an overlap of activation domains for deuterated and non-deuterated odorants (see O2 and O1d domain overlap in Fig. 7), thereby making odorant 2 receptors to become activated by deuterated odorant 1.
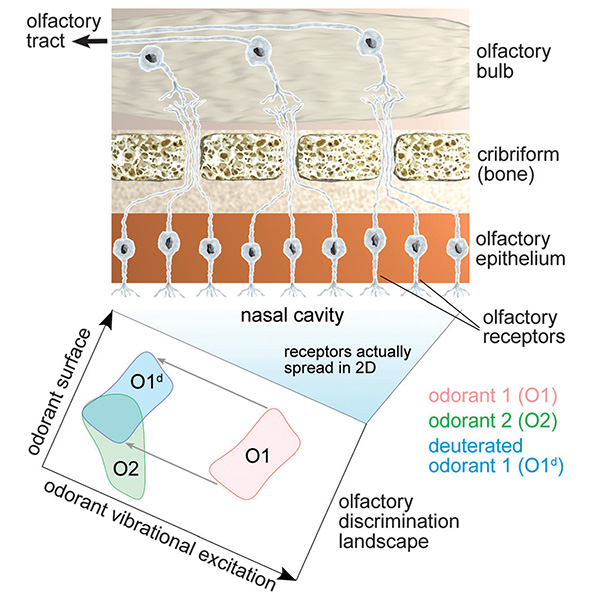 |
Figure 7. Olfactory discrimination landscape. (top) Schematic illustration of a part of the olfactory system consisting of olfactory epithelium, cribriform and olfactory bulb, responsible for detecting different odors. The olfactory receptors shown aligned in a row are actually spread over the two-dimensional nasal cavity. The cavity, through the different receptor properties, becomes an olfactory discrimination landscape. (bottom) Schematic illustration of the olfactory discrimination landscape demonstrating odorant recognition through a combination of surface properties and vibrational excitations. Each odorant, a point in the x,y-plane surrounded by a neighborhood, is attributed to an activation domain on this landscape, as illustrated for two examples (red and green domains). Odorant deuteration corresponds to a domain-shift along the odorant vibrational excitation coordinate (blue domain). |
TCB Publications
Vibrationally assisted electron transfer mechanism of olfaction: Myth or reality? Ilia A. Solov'yov, Po-Yao Chang, and Klaus Schulten Physical Chemistry - Chemical Physics, DOI:10.1039/C2CP41436H, 2012.
Investigators
- Ilia Solov'yov
- Po-Yao Chang
- Klaus Schulten
maintained by Ilia Solov'yov



