Lung Collectins
Overview
Lung surfactant is produced in mammalian lungs and serves a dual role of maintaining normal breathing and shielding the organism from inhaled pathogens. Lung surfactant is a complex mixture of 90% lipid and 10% protein. The most abundant component in surfactant is dipalmitoylphosphatidylcholine (DPPC) lipid, which accounts for 40% of the total mass of lung surfactant. DPPC is essential to forming an air-liquid interface in the lung, and minimizing the contact between hydrophobic region (air) and water molecules, thereby lowering the lung surface tension and preventing lung collapse.
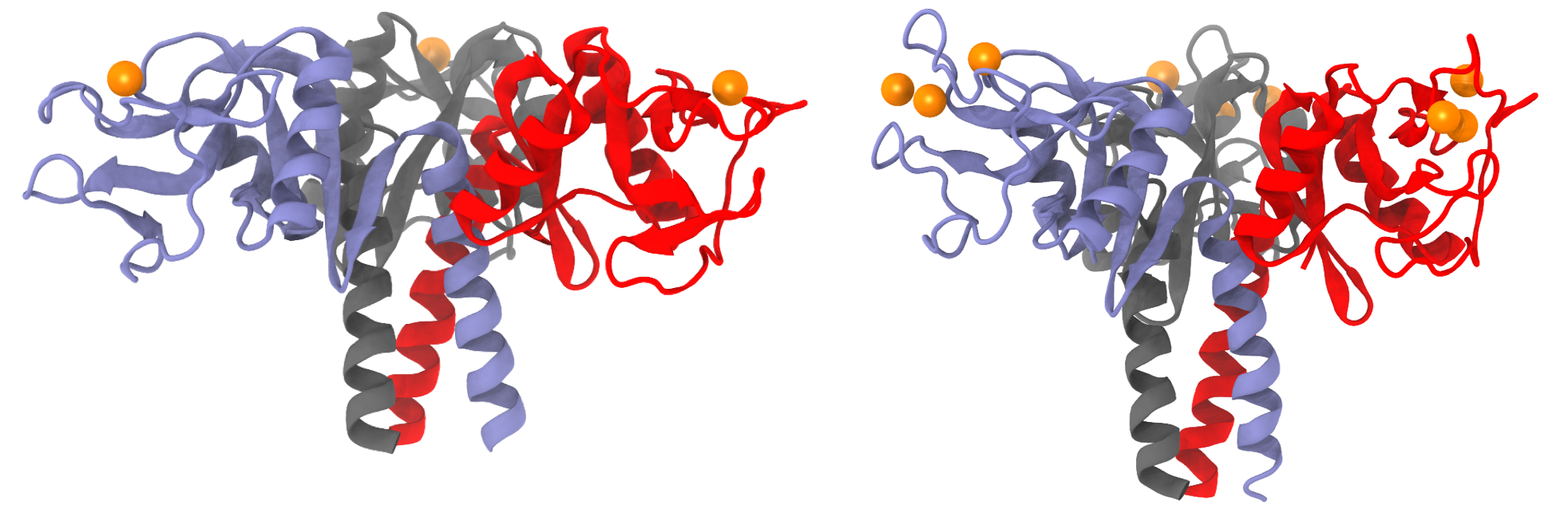
X-ray structures of lung surfactant proteins. SP-A (left) adopts a "T"-shape conformation while SP-D (right) adopts a "Y"-shape conformation. Surfactant proteins are shown as cartoon representations in blue, red, gray colors. Calcium ions at the lectin site are represented as orange spheres.
Surfactant proteins A (SP-A) and D (SP-D) are hydrophilic proteins that belong to the collectin (collagen-containing C-type lectin) protein family, and have antiviral and antimicrobial properties. C-type lectins are carbohydrate-binding proteins that bind to the sugar molecules on pathogenic surfaces using calcium ions. As both SP-A and D use calcium ions to bind sugar molecules, we will refer to the C-type lectin binding site as simply the lectin site. Figure besideshows the slight difference in the overall shapes of SP-A and SP-D. Owing to its "T"-shape conformation, SP-A binds well to flat surfaces like the lipid membrane, making SP-A primarily an antimicrobial protein. On the contrary, SP-D is an antiviral protein, partly due to the more extended "Y"-shape conformation.
Lung surfactant is produced in mammalian lungs and serves a dual role of maintaining normal breathing and shielding the organism from inhaled pathogens. Lung surfactant is a complex mixture of 90 % lipid and 10 % protein. The most abundant component in surfactant is dipalmitoylphosphatidylcholine (DPPC) lipid, which accounts for 40% of the total mass of lung surfactant. DPPC is essential to forming an air-liquid interface in the lung, and minimizing the contact between hydrophobic region (air) and water molecules, thereby lowering the lung surface tension and preventing lung collapse.
Surfactant proteins A (SP-A) and D (SP-D) are hydrophilic proteins that belong to the collectin (collagen-containing C-type lectin) protein family, and have antiviral and antimicrobial properties. C-type lectins are carbohydrate-binding proteins that bind to the sugar molecules on pathogenic surfaces using calcium ions. As both SP-A and D use calcium ions to bind sugar molecules, we will refer to the C-type lectin binding site as simply the lectin site. Figure beside shows the slight difference in the overall shapes of SP-A and SP-D. Owing to its "T"-shape conformation, SP-A binds well to flat surfaces like the lipid membrane, making SP-A primarily an antimicrobial protein. On the contrary, SP-D is an antiviral protein, partly due to the more extended "Y"-shape conformation.
Medical Problem
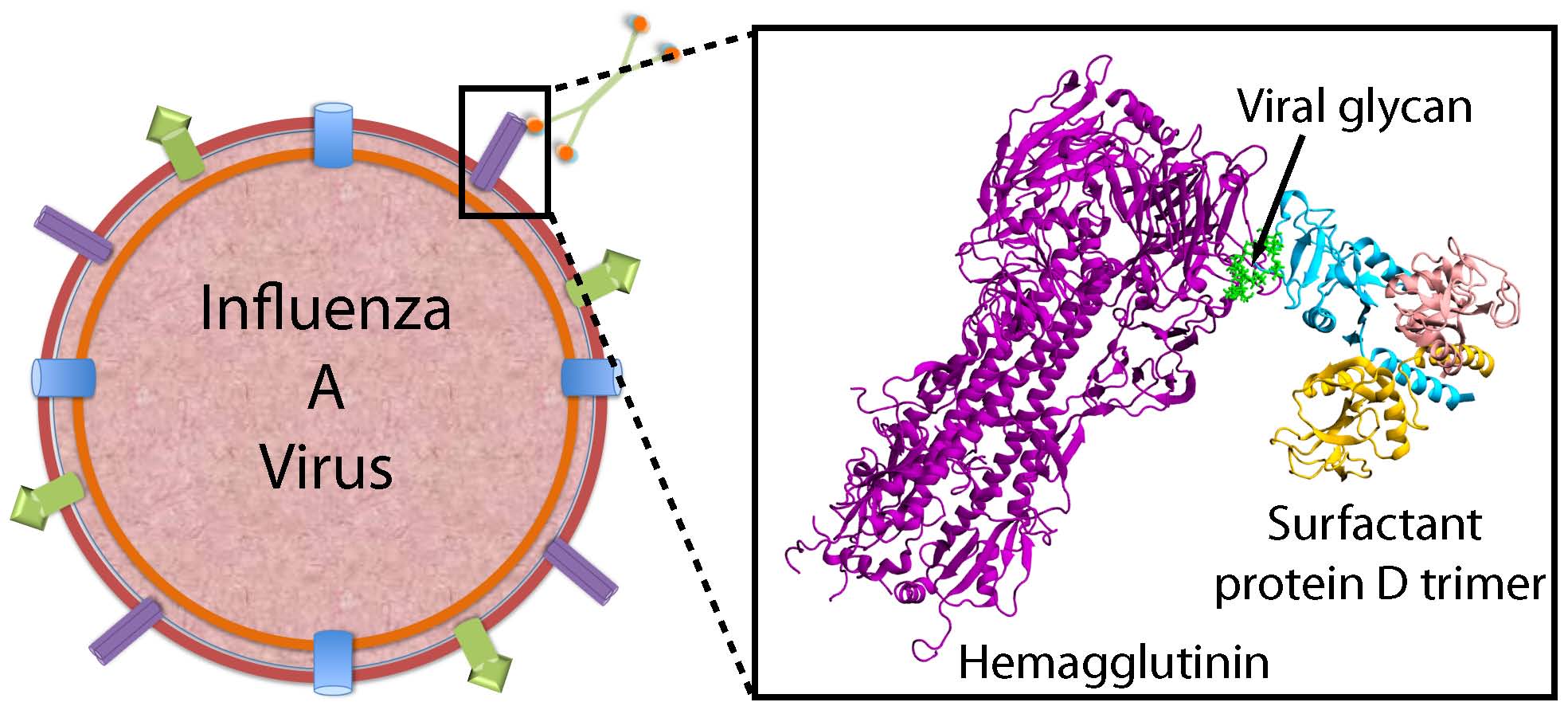
Lung Collectins Defense Against Influenza A Virus
Computational Problems
Accumulating evidence suggests that evasion of collectin recognition is a factor in the severity of pandemic IAV strains. This collaboration will exploit innate, collectin-mediated immune responses against IAV to develop new therapeutic approaches that target new or re-emerging strains of influenza. Our focus is to determine the molecular basis of recognition of inhaled pathogens by surfactant proteins A and D. The following are major questions that need to be addressed. (1) What are the SP-D-HA binding modes and how do the composition and location of HA glycans affect this binding? (2) How do gain-of-function collectin mutations affect IAV binding and neutralization? (3) What are the binding interactions between HA and sialylated collectins like SP-A?
Pig SP-D has a Higher Anti-influenza Activity than Human SP-D
Experimental Observations
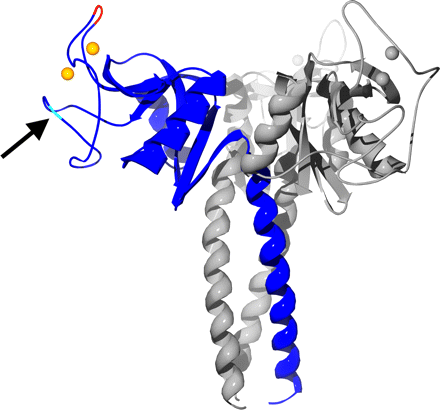
Ribbon diagram of the pig SP-D trimer.
Crystal Structure of pig SP-D trimer
SP-D is mainly secreted as a dodecameric four-armed structure in which each arm represents a trimeric oligomer, the basic structural unit common for the collectin family members. Within each subunit, four major domains can be distinguished as follows: an N-terminal cross-linking domain; a collagen-like triple helical domain; a neck region, and a carbohydrate recognition domain (CRD). The neck region, a short stretch of 33 amino acids, mediates formation of a triply wound alpha-helical coiled-coil domain. The three globular C-type lectin domains are clustered at the C terminus of each subunit to facilitate multivalent high affinity interactions between the ligand-binding sites of SP-D with patterns of glycoconjugates expressed on the surface of pathogens. Prior in vitro and in vivo studies have shown that the trimeric arrangement and carbohydrate-binding characteristics of the CRD are important structural requirements that confer the strong antiviral activity of SP-D against IAV.
The pig SP-D consists of a core with seven beta-sheet regions flanked by two alpha-helices. Two loops on the protein, termed (as in mannan-binding lectin) as the short loop (residues 297-303 in pSP-D) and the long loop (residues 307-331 in pSP-D), located in the CRD distal to the trimer axis, are involved in binding the two calcium ions observed in the structure. In the left image, one monomer is highlighted in blue, and the residues of the insertion at residue 326 is highlighted in red. Calcium ions are shown as orange or gray spheres. The location of the N-glycosylation site is indicated by an arrow.
Lectin site of pig SP-D and human SP-D
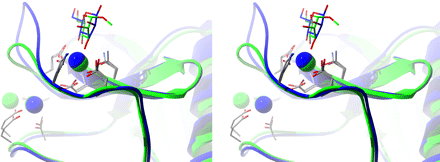
Lectin site of pig SP-D and human SP-D.
The lectin calcium site in RpNCRD-dNG is formed from residues on the long loop as well as residues from a nearby beta-sheet region (residues 339-343) (left figure). The calcium ion is coordinated by six protein oxygen atoms from five amino acids and two oxygen atoms from the ligands, creating a square antiprism. Mannose binds via the O3 and O4 oxygen atoms that form the canonical vicinal equatorial diol recognized by this type of lectin. Two of the amino acids, Glu-321 and Asn-323, are from a consensus sequence EPN found in most C-type lectins that favor binding of mannose. Two other amino acids, Asn-341 and Asp-342, are part of a consensus WND sequence found in C-type lectins. The other amino acid is Glu-329, which coordinates this calcium through its side chain.
A second nonlectin calcium is bound in the site between the two loops and is thought to play a structural role. This calcium is coordinated by five protein oxygen atoms from four protein side chains and one backbone oxygen atom, making a square pyramidal coordination sphere. Two of these side chains are from the short loop Asp-297 and Glu-301, the latter of which makes a bidentate interaction with calcium through its side chain. Other coordination interactions are formed with residues from the long loop, i.e. the side chains of Asn-324 and Asn-330 and the backbone oxygen atom of Glu-329.
Results from Molecular dynamics simulations
We have shown that porcine SP-D exhibits stronger anti-IAV activity than human SP-D, and our studies investigate the reasons for this enhancement. Crystallographic analysis of the trimeric neck-CRD fragment derived from pSP-D complexed with D-mannose shows that compared with the corresponding fragment from hSP-D, a tripeptide insertion in pSP-D slightly alters local conformation at the lectin site. Molecular dynamics simulations show that the loop region (residues 307-331) containing the insertion is flexible in the unliganded form, but the conformation is stabilized upon sugar binding. The simulations further suggest that the loop region could facilitate stronger binding to viral glycans through contact with distal portions of the branched mannoside. The data suggest that differences between the loop region of pSP-D and that of hSP-D are responsible for enhanced IAV antiviral properties of unglycosylated pSP-D relative to hSP-D. The potential relevance of these findings to subclinical IAV infection and viral reassortment in pigs is discussed.
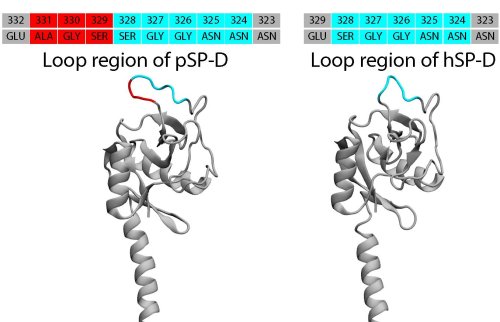
Porcine SP-D (pSP-D) has a SER-GLY-ALA tripeptide insertion (highlighted in red) in the loop region (glycan binding region) compared with human SP-D (hSP-D).
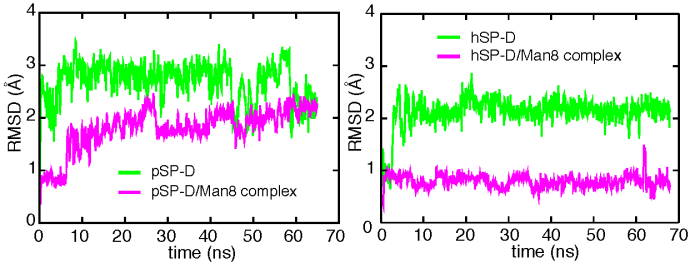
Shown is the time evolution of the RMSD of the loop region of pig and human SP-D (green) and their SP-D/Man8 complex (pink) obtained from molecular dynamics simulations.
Movies
pspd_highlightloop.mpg (3.1MB): The fluctuation of the long loop of pig SP-D (green) in a 65-ns simulation.
pspd_man_highlightloop.mpg (3.9MB): The fluctuation of the long loop of the pig SP-D/Man8 complex (magenta) in a 65-ns simulation. Octamannose is shown in transparent representation.
Enhancing Human SP-D's Anti-influenza Activity via Mutations
The major IAV target for SP-D is the viral glycoprotein hemagglutinin (HA), a major virulence factor that packs densely on the viral surface. HA plays a central role in influenza infection. It is responsible, through its sialic acid receptor site, for attaching IAV to sialylated glycoproteins on host cells and facilitating entry of the viral genome. HA on its own surface expresses high-mannose glycans, particularly a highly branched nonamannose (Man9), which are recognized by SP-D.
We have used X-ray crystallography, molecular modeling, and large-scale molecular dynamics to probe the interactions between SP-D and HA at the atomic level. We compared two forms of human SP-Ds: wild type (WT) and double mutant D325A+R343V. This study, which offers the first computational investigation involving a complex of a full hemagglutinin trimer and a SP-D trimer, has allowed us to investigate the detailed molecular interactions between SP-D and IAV HA.
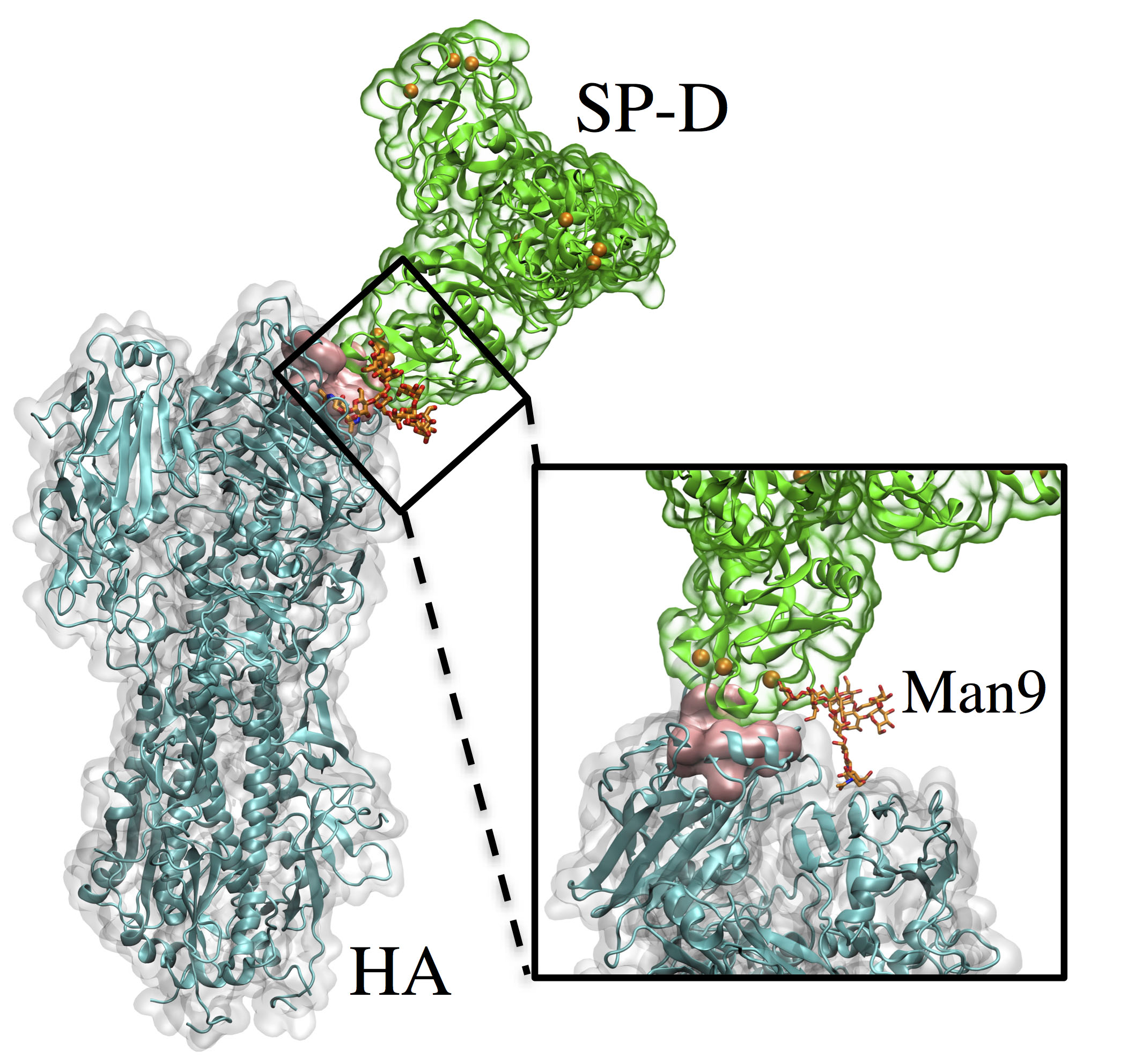
View of the entire simulated system. Hemagglutinin (HA) and lung surfactant protein-D (SP-D) are connected noncovalently through a Man9 chain, shown enlarged in the inset with the sialic acid binding site (pink surface), and the calcium ions of SP-D are shown as orange spheres.
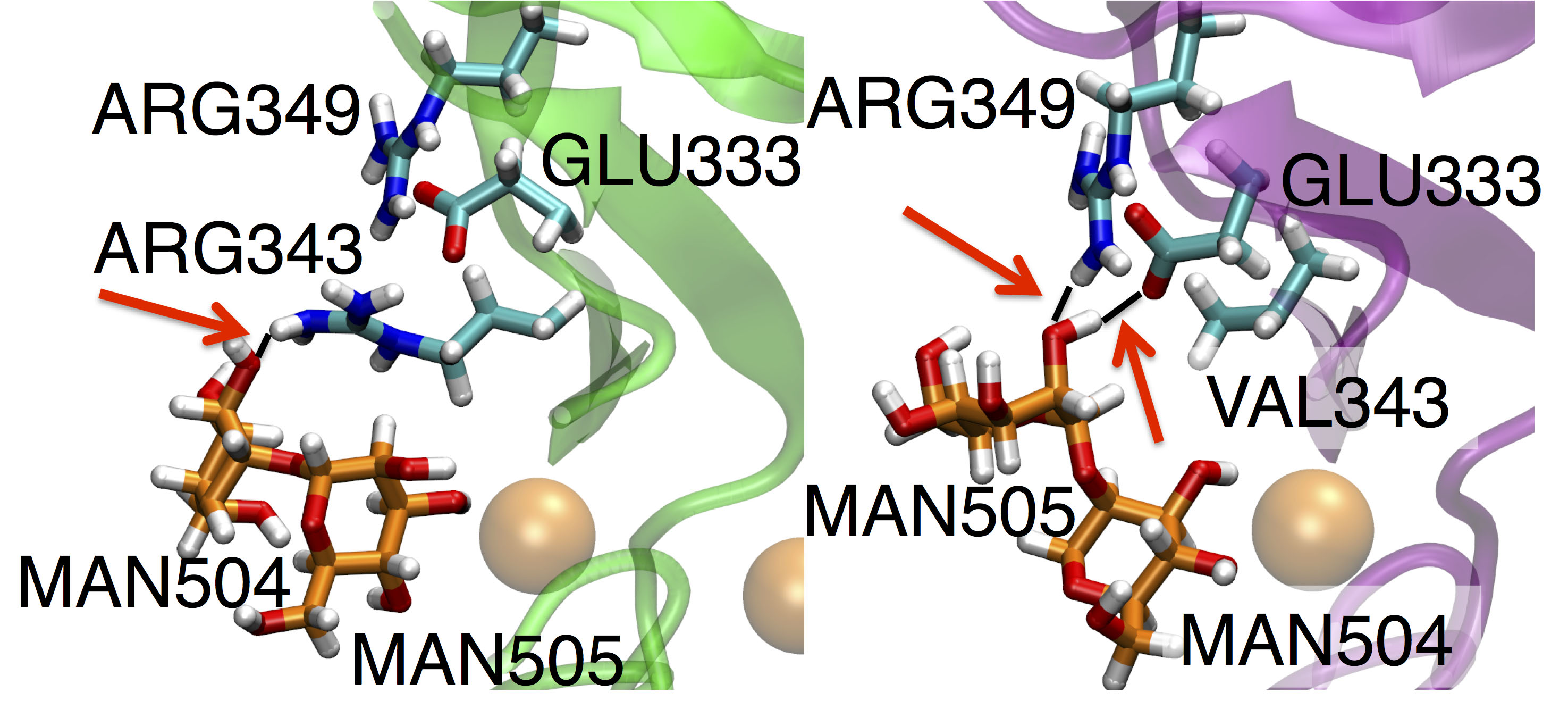
R343V mutation alters the Man9 binding mode. Shown is the difference in hydrogen bond (red arrows) networks for the Man9 chain binding to WT (left) and D325A+R343V (right).
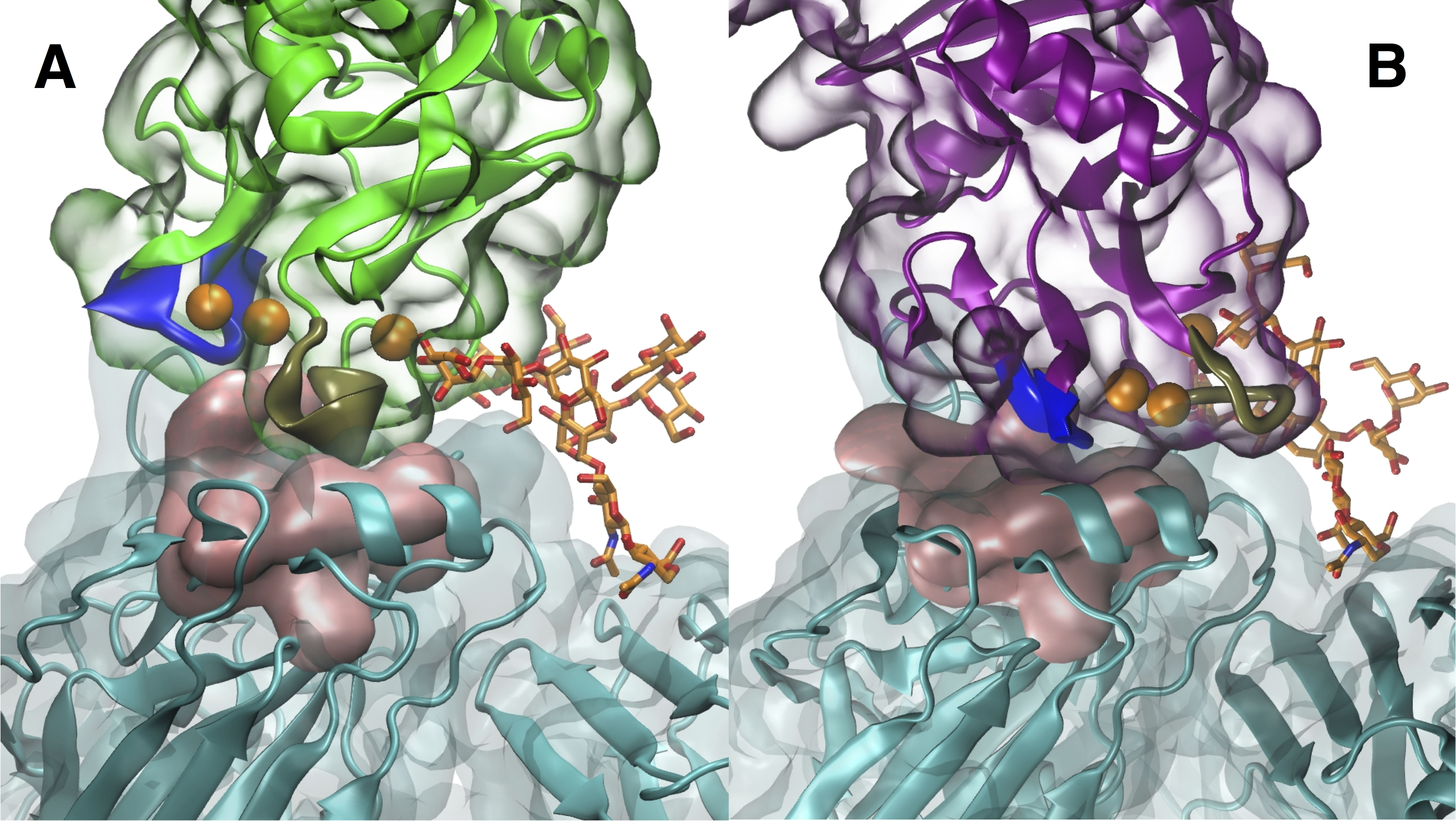
Inhibition of the sialic acid binding site. (A) WT SP-D blocks the binding site with a tan loop while (B) D325A+R343V uses its blue loop to cover and thereby inhibit the sialic acid binding site.
Using modeling and molecular dynamics approaches, we have determined that the double mutant SP-D not only binds stronger to the glycan chains on HA but also blocks the SA binding site better than WT SP-D upon binding to the glycan chains. The altered binding orientation and increased level of viral aggregation, coupled with the increased level of binding to HA by the double mutant, enhance the ability of D325A+R343V to neutralize IAV. These studies thus shed light on how factors associated with HA binding correlate with antiviral activities of SP-D.
Host Defense Properites of SP-A
Like SP-D, SP-A plays an important role in pulmonary innate immunity by recognizing canonical patterns on microbial surfaces. It protects the lungs from infection by recognizing the lipid component (lipid A) on gram negative bacterial surfaces, and by helping to initiate various clearance mechanisms. SP-A is also known to aggregate dipalmitoylphosphatidylcholine (DPPC), the major constituent of lung pulmonary surfactant, to form lamellar bodies and tubular myelin, a highly structured form of surfactant lipids and proteins. It is unclear how SP-A can bind to both DPPC and lipid A for two very different functions.
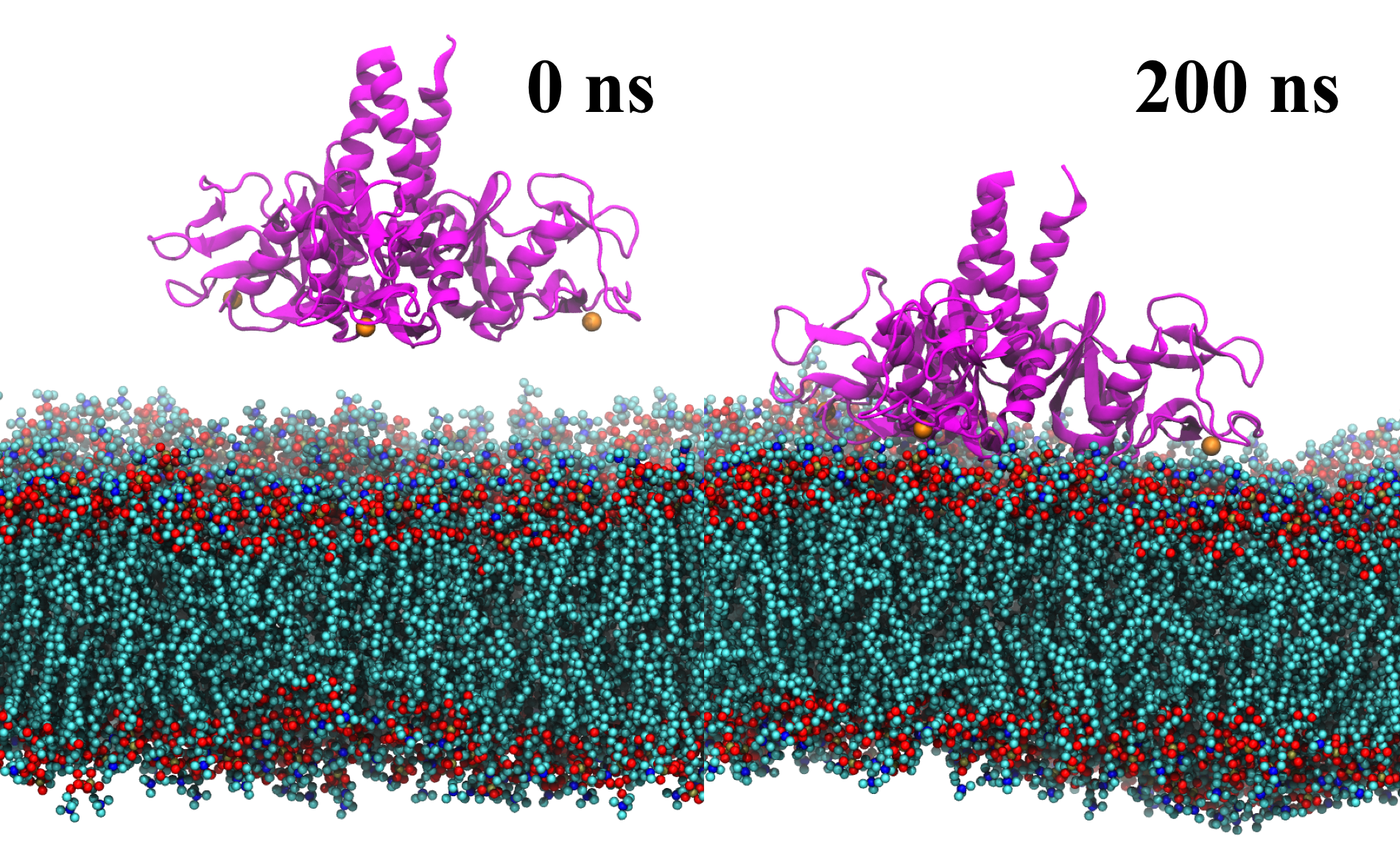
Lipid membrane binding by SP-A simulated using MD simulations. SP-A (ribbon representation colored magenta) diffuses and binds firmly to the membrane bilayer within 150 ns and stayed bound for an additional 50 ns.
To unravel the dual role of SP-A, we performed crystallographic and mutational analyses as well as all-atom MD simulations. In previous crystallographic studies of SP-A, we studied a construct containing the entire CRD and neck domain (NCRD), which recapitulates many of the binding properties of the intact protein. In the initial report, we observed that the tyrosine-rich CRD surface included three surface-exposed tyrosine residues (Y164, Y208, and Y192) arranged in part as a box-like structure, and a proximal basic cluster containing two arginines (R216 and R222). These residues were hypothesized to form part of a phosphocholine binding site via an apparent pi-cation interaction and hydrogen bonds. The validity of the phosphocholine site were further tested using mutagenesis and binding assays.
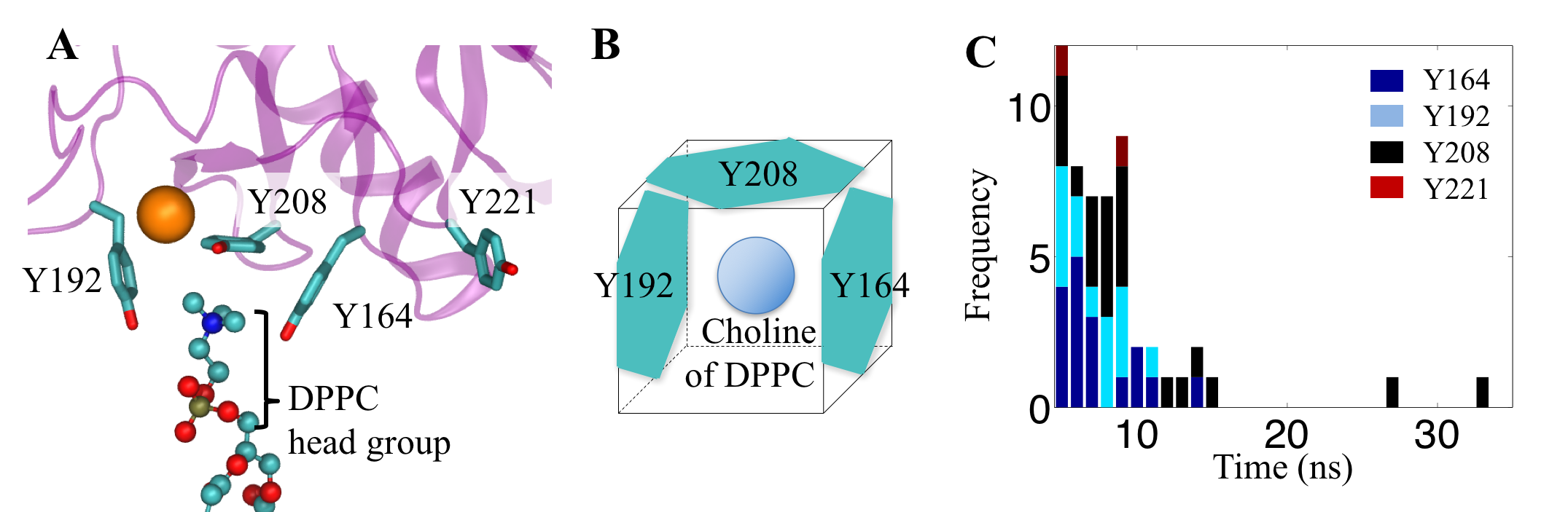
Tyrosine box that amplifies the cation-pi interaction. (A and B) Three tyrosine residues of SP-A form a box-like conformation to bind with choline, part of the DPPC lipid headgroup. (C) histogram showing the duration of a choline bound to the key tyrosine residues of SP-A.
Finally, we extended these studies to investigate SP- A/lipid A complexes, for which there are no crystallographic data, utilizing large-scale molecular dynamics simulations to investigate DPPC and lipid A binding in a membrane milieu. MD simulations also revealed that SP-A binds to lipid A more tightly than to DPPC. These results suggest that SP-A may transfer from surfactant DPPC to pathogen membranes to initiate its host defense functions. These combined studies present a cohesive view of the molecular interactions, on an atomic level, of SP-A and two of its major lipid ligands as occur in the presence of membranes.
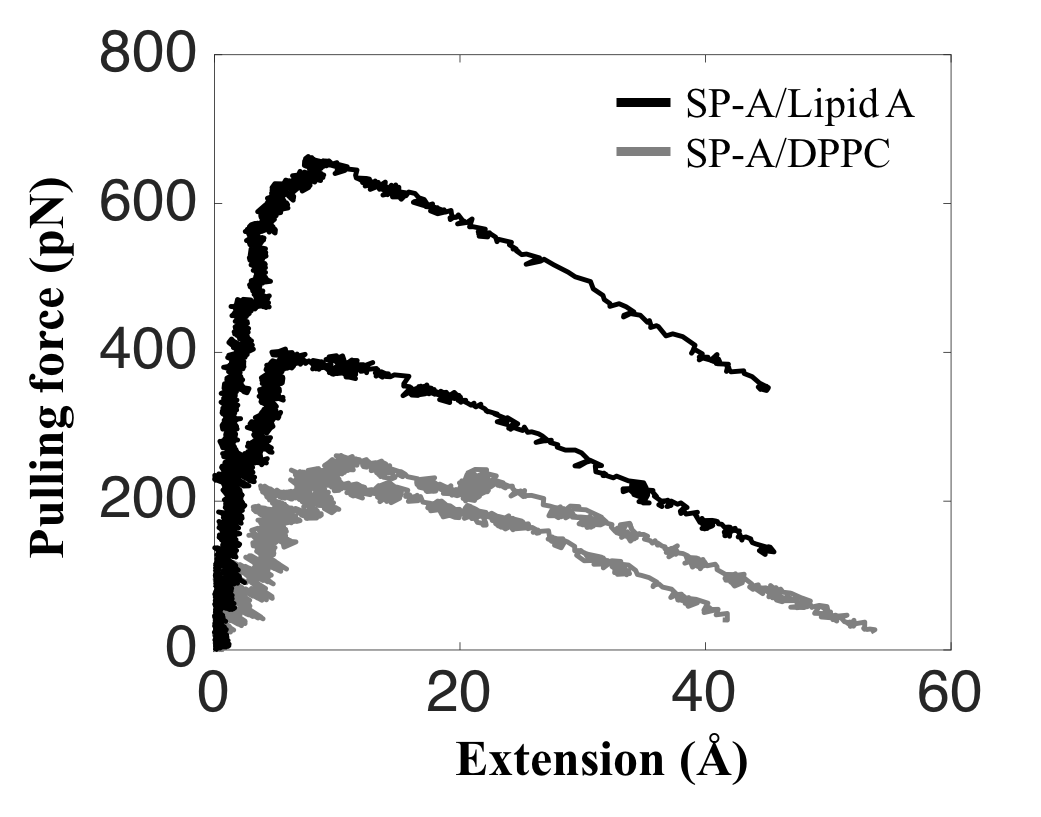
Larger force is required to pull SP-A off a lipid A than off a DPPC membrane. The force-extension curves for SP-A/Lipid A (black) show that the applied pulling forces are at least 60% larger than that of SP-A/DPPC (gray).
Publications
Elucidation of Lipid Binding Sites on Lung Surfactant Protein A Using X-ray Crystallography, Mutagenesis, and Molecular Dynamics Simulations
Boon Chong Goh, Huixing Wu, Michael J. Rynkiewicz, Klaus Schulten, Barbara A. Seaton, and Francis X. McCormack. Biochemistry, 55:3692-3701, 2016Molecular mechanisms of inhibition of influenza by surfactant protein D revealed by large-scale molecular dynamics simulation.
Boon Chong Goh, Michael J. Rynkiewicz, Tanya R. Cafarella, Mitchell R. White, Kevan L. Hartshorn, Kimberly Allen, Erika C. Crouch, Oliviana Calin, Peter H. Seeberger, Klaus Schulten, and Barbara A. Seaton. Biochemistry, 52:8527-8538, 2013.Unique sugar-binding site mediates the distinct anti-influenza activity of pig surfactant protein D.
Martin van Eijk, Michael J. Rynkiewicz, Mitchell R. White, Kevan L. Hartshorn, Xueqing Zou, Klaus Schulten, Dong Luo, Erika C. Crouch, Tanya M. Cafarella, James F. Head, Henk P. Haagsman, and Barbara A. Seaton. Journal of Biological Chemistry, 287:26666-26677, 2012
Investigators
Page created by Xueqing Zou, and maintained by Boon Chong Goh.



