Highlights of our Work
2024 | 2023 | 2022 | 2021 | 2020 | 2019 | 2018 | 2017 | 2016 | 2015 | 2014 | 2013 | 2012 | 2011 | 2010 | 2009 | 2008 | 2007 | 2006 | 2005 | 2004 | 2003 | 2002 | 2001
Biological cells, in particular neurons, maintain an inside-outside
voltage gradient through active transport of ions (Na+, K+,
Cl-, and others) across their membranes. The flow of the ions
down their gradients through membrane channels is highly selective for
each ion. The high selectivity permits nerve cells to signal each
other through voltage spikes, which are produced through
transient changes of channel conductivities for Na+ ions (channels
open and close in about a ms) and K+ ions (channels open and close in
about 10 ms). Crucial for the generation of voltage spikes is the
selective, yet quick, conduction of ions, but as one knows
from personal experience at border crossings, high selectivity and quick crossing
seem to be mutually
exclusive. Yet biological ion channels reconcile selectivity and
speed. Prior experimental work, primarily that of year 2003 Nobelist
MacKinnon, as well as computational work suggested how potassium
channels achieve selectivity and speed.
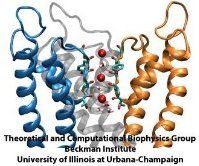
But until recently no high resolution atomic structure of a potassium
channel was known in the open form and the suggested mechanism could
not be tested under natural conditions through atomic level
simulations. Last year's solution of the
structure of the potassium channel Kv1.2 in its open form made it
finally possible to simulate, using NAMD, the conduction of ions
through Kv1.2 driven by a voltage gradient. The results reported
recently confirmed indeed the high selectivity - high speed mechanism
suggested earlier, namely a billiard-type motion of two and three
ions, the last ion kicking the first ion out. The
simulations revealed for the first time, through movies, the overall
permeation process, including the jumps of ions between energetically
favorable binding sites and the sequence of multi-ion configurations
involved in permeation. More on our
potassium channel web site.